By Sneha S K and Sahil Pandey
Dec 31 (Reuters) - The U.S. Food and Drug Administration has declined to approve Corcept Therapeutics' drug for the treatment of a rare hormonal disorder, the company said on Wednesday.
Shares of the drugmaker were down 48% at $36.41.
The company said the FDA could not arrive at a favorable benefit-risk assessment for the hormone-blocking oral treatment, known as relacorilant, without Corcept providing additional evidence of effectiveness.
The company was seeking approval for relacorilant as a treatment for patients with hypertension secondary to hypercortisolism.
"FDA's request for additional data may require additional trials, significantly dimming Corcept's outlook in Cushings," said Truist analyst Joon Lee.
Hypercortisolism, also known as Cushing's syndrome, occurs when the body is exposed to high cortisol activity.
Corcept had submitted trial data that showed that relacorilant made improvements in a wide array of hypercortisolism's signs and symptoms.
"We will meet with the FDA as soon as possible to discuss the best path forward," said Joseph Belanoff, Corcept's CEO.
Main symptoms of hypercortisolism include a fatty hump between the shoulders, a rounded face, and pink or purple stretch marks on the skin. People with Cushing's also experience diabetes, high blood pressure, muscle weakness and immune suppression.
Relacorilant is a selective cortisol modulator designed to block the effects of cortisol, while avoiding certain off‑target hormonal effects.
"Given the company had opportunities to address FDA's concerns during mid and late-stage reviews, it's unclear if any further dialogue can resolve the review issues without additional trials," Lee added.
Corcept is also studying the drug in a variety of serious disorders including ovarian and prostate cancer. Its other drug known as Korlym is approved to treat high blood sugar caused by hypercortisolism in adults with endogenous Cushing's syndrome.
Other approved treatments for Cushing's syndrome include Isturisa by Recordati and Xeris Biopharma's Recorlev.
(Reporting by Sahil Pandey and Sneha S K in Bengaluru; Editing by Shailesh Kuber)
latest_posts
- 1
 6 Novice Cameras for 2024: Ideal for New Picture takers
6 Novice Cameras for 2024: Ideal for New Picture takers - 2
 Bold Colors, Playful Shapes, Handmade Textures: This Mexico City Home Is 100% Joyful!
Bold Colors, Playful Shapes, Handmade Textures: This Mexico City Home Is 100% Joyful! - 3
 Kelsey Grammer on having a new baby at 70: 'You're just more available now'
Kelsey Grammer on having a new baby at 70: 'You're just more available now' - 4
 Culinary Joys: Investigating Connoisseur Cooking at Home
Culinary Joys: Investigating Connoisseur Cooking at Home - 5
 New movies to watch this weekend: See 'We Bury the Dead' in theaters, rent 'Wicked: For Good,' stream 'The Unbreakable Boy' on Starz
New movies to watch this weekend: See 'We Bury the Dead' in theaters, rent 'Wicked: For Good,' stream 'The Unbreakable Boy' on Starz
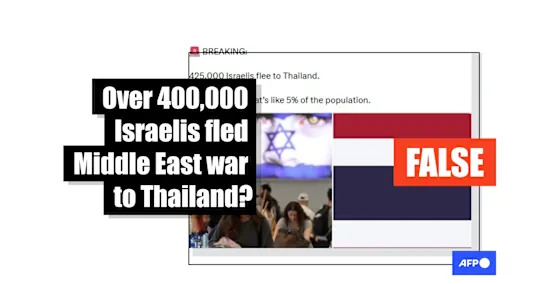
 Israeli tourist data from 2025 misrepresented as mass exodus to Thailand
Israeli tourist data from 2025 misrepresented as mass exodus to Thailand There are thousands of aligned holes in Peru. Archaeologists now think they know who made them
There are thousands of aligned holes in Peru. Archaeologists now think they know who made them Hezbollah claims right to respond to killing of top commander
Hezbollah claims right to respond to killing of top commander JFK's granddaughter reveals terminal cancer diagnosis, criticizes cousin RFK Jr.
JFK's granddaughter reveals terminal cancer diagnosis, criticizes cousin RFK Jr. Tourist trade in Greece and Cyprus suffering from Iran war effects
Tourist trade in Greece and Cyprus suffering from Iran war effects Shooting of MIT professor Nuno Loureiro has police searching for a suspect
Shooting of MIT professor Nuno Loureiro has police searching for a suspect Virtual National Science Foundation internships aren’t just a pandemic stopgap – they can open up opportunities for more STEM students
Virtual National Science Foundation internships aren’t just a pandemic stopgap – they can open up opportunities for more STEM students Coca-Cola Co. and bottlers to invest in South Africa operations
Coca-Cola Co. and bottlers to invest in South Africa operations Police break up illegal chicken slaughter in Germany
Police break up illegal chicken slaughter in Germany